
An Introduction to the Physics of Scuba Diving
Scuba diving is a captivating adventure that unveils a realm of beauty and mystery beneath the ocean’s surface. It’s a world where gravity seems to lose its grip, where colourful marine life dances amidst the currents.
Yet behind the wonder lies a complex interplay of physics principles that govern every dive. From Boyle’s Law to Dalton’s Law, each concept plays a vital role in ensuring the safety and success of divers as they venture into the depths.
Boyle’s Law
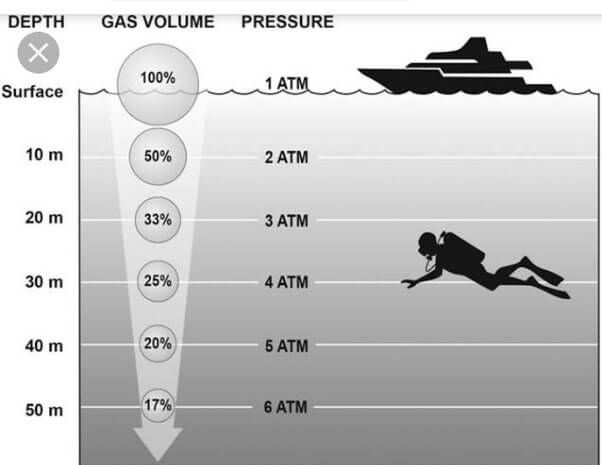
Boyle’s Law, named after the Irish physicist Robert Boyle, explains the relationship between the pressure and volume of a gas at a constant temperature. Simply put, as pressure increases, volume decreases and vice versa. This law has significant implications for scuba divers, especially as they descend into the depths.
Imagine a diver descending into the ocean holding onto a balloon. The balloon will shrink under pressure when submerged just as the volume of air in the diver’s tank decreases with depth due to increased pressure. At a depth of 30 meters, the pressure is approximately four times greater than at the surface, causing the air volume in the tank to shrink accordingly. This reduction in volume highlights the importance of monitoring air supply to ensure a safe return to the boat or sure as we ascent back to the surface. This is also why we need to add and subtract air from our BDC as we ascend and decent to keep our buoyancy constant.
Boyle’s Law also comes into play when divers conduct technical dives at greater depths, where they may encounter different gas mixtures such as trimix to mitigate the effects of nitrogen narcosis. Understanding how changes in pressure affect gas volume is crucial for selecting the appropriate gas mixtures and ensuring the safety of deep-water exploration.
Moreover, Boyle’s Law extends beyond the realm of scuba diving. It’s fundamental in various fields such as aerospace engineering where it’s used to design and test aircraft cabins, ensuring passengers’ comfort and safety at different altitudes. Similarly in medicine, Boyle’s Law is applied in respiratory therapy to regulate the flow of gases into the lungs.
Charles’s Law
Charles’s Law, named after the French scientist Jacques Charles describes the relationship between the volume and temperature of a gas at constant pressure. As temperature increases, volume expands and as temperature decreases, volume contracts. This principle is crucial for divers as they encounter temperature variations during their dives.
Picture a diver ascending from the chilly depths of a night dive to the warmer surface waters. As the temperature rises the gas in their buoyancy control device (BCD) expands, providing them with additional buoyancy. This change in buoyancy affects the diver’s ability to maintain a stable ascent. By understanding Charles’s Law, divers can anticipate these fluctuations and make necessary adjustments to their equipment to ensure a smooth transition between depths.
Furthermore, Charles’s Law finds application in everyday scenarios, such as inflating a balloon. When you blow air into a balloon and then expose it to heat the air inside expands, causing the balloon to inflate further. Conversely, if you place the balloon in the freezer the cold temperature causes the air inside to contract, resulting in a deflated balloon. This concept mirrors the behaviour of gases underwater, where temperature changes affect the volume of gas in a diver’s equipment.
Henry’s Law
Henry’s Law governs the solubility of gases in liquids and is of particular importance for understanding decompression sickness in divers. As a diver descends the pressure increases, meaning gases like nitrogen and oxygen dissolve in their bloodstream. Ascending too quickly can lead to the formation of bubbles in the bloodstream as the gas rapidly expands, causing decompression sickness.
Imagine opening a bottle of soda and observing the bubbles fizzing up as you open the lid – the pressure is decreasing. This analogy illustrates Henry’s Law in action, where the dissolved gases come out of solution due to the change in pressure. Similarly, as divers ascend from the depths the decreasing pressure causes dissolved gases in their bloodstream to form bubbles, potentially leading to decompression sickness if not managed properly. This is why we surface slowly from every dive and you slowly unscrew your soda lid.
A comprehensive study published in the “Journal of Physiology” (Vol. 589, Issue 6) delves into the mechanisms of decompression sickness, emphasizing the role of Henry’s Law in gas exchange and bubble formation in divers.

Dalton’s Law
Dalton’s Law describes the behaviour of gas mixtures, stating that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures of each individual gas in the mixture. Therefore as pressure increases so does partial pressure and visa versa. This principle guides divers in selecting appropriate depth limits for gas mixtures such as Nitrox 32.
Consider a diver exploring a deep underwater wreck. To extend his exposure to nitrogen and increase his bottom time he breathes a gas mixture with a lower nitrogen content, such as Nitrox 32. Nitrox 32 is a mixture composed of 32% oxygen and 68% nitrogen. Both of these gases can have negative impacts on a diver at high pressures, with oxygen being particularly toxic. Symptoms of oxygen toxicity can include changes in vision, dizziness/vertigo, and seizures, all of which can be problematic for a diver and can lead to death. Using Dalton’s Law we can calculate at what depth a diver might begin to experience symptoms of oxygen toxicity when diving and easily avoid those depths.
Using Dalton’s Law, divers can safely navigate the complexities of deep water environments.
Moreover, Dalton’s Law has practical applications beyond scuba diving, such as in the production of industrial gases. In manufacturing processes where specific gas mixtures are required, understanding Dalton’s Law ensures the precise control and regulation of gas compositions to meet desired specifications.

Conclusion
In essence, scuba diving physics serves as the foundation for safe and enjoyable underwater exploration. By embracing principles like Boyle’s Law, Charles’s Law, Henry’s Law and Dalton’s Law, divers can dive with confidence, knowing they have a solid understanding of the forces at play beneath the waves. These fundamental principles supported by scientific research and practical applications, enhance the safety and success of divers as they navigate the mesmerizing depths of the ocean.
From managing gas volumes to anticipating temperature fluctuations, these principles guide divers through every aspect of their underwater journey. By embracing the fundamental laws of physics, divers can unlock the wonders of the ocean while ensuring their safety and well-being.


